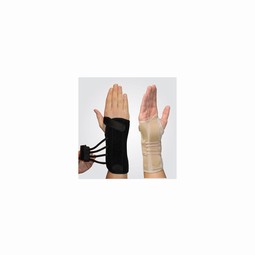
Prisma - Stabil Plus
Extra stable wrist bandage with malleable bars volart. 2 removable spiral springs located dorsally. Adjustable thumb grip. Ulnar rail pocket which allows for extra support. 2 plastic rails included for less rigid support. Lace closure for easy installation Stable and durable material: Colour Black length 18 cm size XS-XXL
Classification
06 06 12 02 - Wrist orthoses
The product series contains 1 product.
Product 1 of 1
Prisma - Stabil Plus
HMI-no.
129727
Article-no.
7070
Registration date
21-06-2021
Last updated
13-08-2024
Properties
With action on the wrist
Yes
With action on the base of the thumb
No
With baleens
Yes
Elastic material
No
Test information
No information about tests according to standardsThe supplier has not provided documentation, that this product is testet according to a relevant standard. See examples of relevant standards for this product type
EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
Other specifications
material: Stable Plus: material with padded inside. Made of cotton, polyester and acrylic (latex free). Size Circumference around the wrist xx-small 10 - 12 cm x- small 12 - 14 cm Small 14 - 16 cm Medium 16 - 18 cm Large 18 - 20 cm X-Large 20 - 22 cm XX-Large 22 - 24 cm
Please state variant when ordering
Please state variant when ordering