All Up Multi Small Silent & Soft
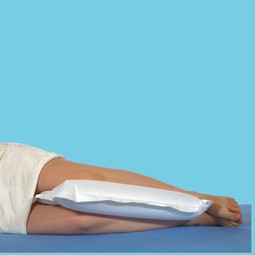
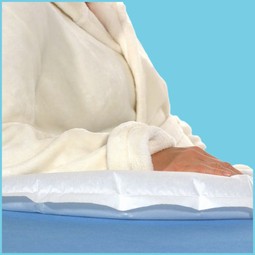
The Multi Small Silent & Soft cushion is a one-patient multifunctional standard cushion that ensures an efficient and even pressure distribution when used for both lying/sitting users. For example, it can be used under the legs, between the knees, behind the back and under the arms.
The pillow is extra soft and silent.
The pillow is extra soft and silent.
Classification
09 07 06 09 - Rectangular positioning cushions
Documents
The product series contains 1 product.
Product 1 of 1
All up Multi Small Silent & Soft - pressure-relieving cushion
HMI-no.
134027
Article-no.
70105
Registration date
17-10-2022
Last updated
05-03-2025
Properties
Filling
Luft
(Optional) machine washable cover
Yes
(Optional) cover which can be autoclaved
No
(Optional) cover which can be disinfected
Yes
Machine washable cushion
Yes
Cushion can be autoclaved
No
Cushion can be tumble dried
No
Flame resistant
No
Measures
Height
8
cm
Longest side
50
cm
Shortest side
25
cm
Test information Though the product is testet, it might not have passed all requirements in the standard. Besides, some products are only tested according to parts of the standard. Read the test report for detailed information. See also other standards that could be relevant for this product type.
: Tested according to other national or international standard.
Test lab: BEO Berlin. Test date: 23-08-2019 Test report for HMI-no. 134027
Test report for HMI-no. 134027
Test lab: BEO Berlin. Test date: 23-08-2019
 Test report for HMI-no. 134027
Test report for HMI-no. 134027EU product safety regulation The supplier has provided the following information about CE-marking of the product. Explain CE-marking
CE marked as medical device
Other specifications
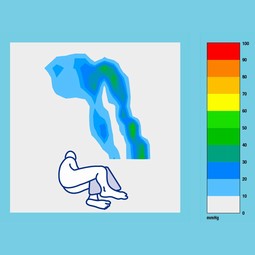
The pad is used for the prevention of pressure ulcers and the relief of pressure ulcers.
All Up Multi Small Silent & Soft is inflatable and covered with nonwoven felt, which is a skin-friendly material that is moisture-absorbent, heat-transporting, soft
All Up Multi Small Silent & Soft is inflatable and covered with nonwoven felt, which is a skin-friendly material that is moisture-absorbent, heat-transporting, soft